All published articles of this journal are available on ScienceDirect.
Investigating Micronucleus Changes in Mouse Lymphocytes Due to Exposure to Silica Dust
Abstract
Background and Objectives:
Occupational exposure to silica dust has multiple consequences, including genetic complications. One of the genetic complications is Micronucleus (MN) changes; therefore, this study aims to evaluate the rate of MN formation in mouse lymphocyte cells due to exposure to silica dust.
Materials and Methods:
Totally 72 male mice BALB/c were selected and categorized into five exposure groups with 12 mice in exposure to the concentrations of 1.3, 3, 8, 12, and 17 mg/m3 of 99% pure silica dust and a control group. The mice were exposed to silica dust in which they were exposed for 8 hours a day, 6 days a week, and for 1, 2, 3, and 4 months. Then, blood samples were taken from the mice and the rate of MN formation in their lymphocyte cells was evaluated. The results were analyzed via SPSS software version 21 (P<0.05).
Results:
Maximum and minimum averages of dust concentration, related to boxes 1 and 5, were 17 mg/m3 and 1.3 mg/m3, respectively. Maximum rate of MN formation was observed in the fourth month of exposure and in group 1 with the value of 21.6±1.15, and minimum rate of MN formation was observed in the third month of exposure and in control group with the value of 3±1. Average of MN frequencies in each of the exposure month was significant related to the control group (P=0.001). There was a direct and significant correlation between exposure concentrations of exposed group and average rate of MN formation (r=0.679).
Conclusions:
More than 3 months exposure to silica dust may lead to significant MN formation in lymphocytes of mice BALB/c in comparison with the control group.
1. INTRODUCTION
Silica is the main mineral on earth and many people are occupationally exposed to it in several industries, such as mine, foundry, ceramic tile factories, production of washing powder and cleaners, brick, production of various chemicals and glazed ceramic tiles, different types of crystals, pharmacy, sand blasting, and ferrosilicon [1]. International Agency for Research on Cancer (IARC) has introduced crystalline silica as a human carcinogen in 1997 [1]. Various studies have been carried out in this field. Several studies evaluate the relationship between diseases and previous exposure with crystalline silica [2-6]. Some toxicological studies were carried out to evaluate health risks of exposure to crystalline silica on human beings [7] and animals [8-10]. Nowadays, it is generally confirmed that biological responses caused by exposure to crystalline silica are under the influence of inherent properties of silica dust and physic-chemical changes. Investigating the effects of silica on genetic content of cells is a new field of research which has been highly focused on in the recent years. In a study by Demircigil et al., on nasal cells and workers’ lymphocytes, it is indicated that dust can form micronucleus (MN) in direct nasal and indirect lymphocyte exposures [11].
| Box Number | Concentration of Dust (mg/m3) | ||||
|---|---|---|---|---|---|
| PM1 | PM2.5 | PM5 | PM10 | Total | |
| 1 | 6.8 | 7.8 | 11.7 | 15.5 | 17 |
| 2 | 4.9 | 5.56 | 8.32 | 10.5 | 12 |
| 3 | 3.1 | 3.7 | 5.74 | 6.59 | 8 |
| 4 | 1.42 | 1.9 | 2.013 | 2.7 | 3 |
| 5 | 0.82 | 1.01 | 1.22 | 1.23 | 1.3 |
MN formation is one of the symptoms of chromosomal aberrations which can be evaluated in low costs and limited facilities and is used to determine chromosomal aberrations due to different physical and chemical factors [12, 13]. Different chemical, physical, and biological factors in living and working environments, such as genotoxic factors, may lead to MN formation [5-10]. Other factors, including temperature above 39oC form MN in sensitive cells [14].
In vitro exposure of human lymphocyte cells with free silica may form more MN in these cells [15]. In a study on the effects of amorphous silica nanoparticles, it is indicated that silica nanoparticles form MN [16]. The studies which have been carried out on crystalline silica at the concentration of 120 mg/ml were cultured on lymphocytes for 24 hours leading to formation of MN; however, no significant difference was observed in their DNA fragmentation with comet assay [15]. Multiple studies indicated that amorphous silica has no genetic and MN complications [17-21]. In a study by Brown et al., two rod and spherical forms of amorphous silica were compared and no significant difference was observed in their silica complications [22].
The main role of the effect biomarkers is to obtain early signs of disease and prevent the development of irreversible results, such as the risk of occupational carcinogens using genotoxicity assays [11]. So far, no valid biomarker has been recommended for early diagnosis and measurements of progression of silicosis [23, 24]. Based on the results of previous studies, MN formation can be regarded as a symptom of chromosomal damage by chemical agents such as crystalline silica [12, 13]. In relation to exposure to crystalline silica dust, there is a need for biomarkers to predict the possibility of silicosis and the development of lung cancer. Advantages of using primary biomarkers from biological effects in etiology studies are that people may be likely less to evaluate the outcome of the disease than cohort studies [11]. This study aims to evaluate the rate of MN formation in mouse lymphocytes due to exposure to silica dust.
2. MATERIALS AND METHODS
This experimental study was carried out on 72 BALB/c male mice in 2016-2017 which were provided from Research Center of Experimental Medicine at Birjand University of Medical Sciences. Average weight of mice was 22 ± 4 g preserved in standard environmental conditions of the laboratory. In the present study, all the ethical issues in relation to working with laboratory animals were considered. For the experiments, five exposure groups with 12 mice in exposure to the concentrations of 1.3, 3, 8, 12, and 17 mg/m3 of silica dust and a control group were selected. The mice were exposed to silica from the sixth week after their birth. The mice were exposed to silica dust respiratory in which they were exposed for 8 hours a day, 6 days a week, and for 1, 2, 3, and 4 months. The silica used in this study was 99% pure which was obtained in Iranian mines, and was used more as sand casting and raw materials in ceramic and glass industries. Dust was created by a blower pump inside a container, and after level out, it was conveyed by the hose into the exposure chambers. Control dampers were used to adjust dust concentration inside the chambers, and an input from each compartment was connected to the dust measuring device to measure dust concentration, and concentration of dust was recorded. Finally, the average concentration was calculated over the period. After exposing the mice with silica dust for 1, 2, 3 and 4 months, blood samples were taken. Nearly 3 cc of blood was taken from the heart of each mouse and blood lymphocytes were separated by centrifuging the blood samples with 3300 rpm for 15 minutes using Ficoll of Sigma Company. The separated lymphocytes were transferred to cell culture laboratory to investigate their MN. MN-contained cells were counted by NIKON microscope. The resulted data were analyzed via SPSS software version 21 (Inc. USA). P<0.05 was considered as significant.
3. RESULTS
Average density and distribution of dust particles in the studied groups are indicated in Table 1. Maximum and minimum dust concentration, related to boxes 1 and 5, were 17 mg/m3 and 1.3 mg/m3, respectively.
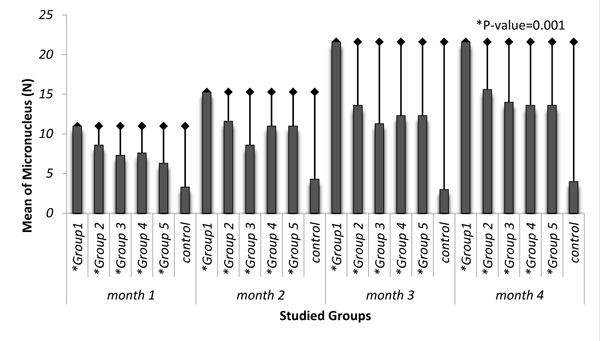
Average changes of MN in mouse lymphocytes of studied groups exposed to silica dust are shown in Table 2. Among the exposure groups, the highest rate of MN formation was in the first group and in the fourth month of exposure. The average number of formed MN in all groups was statistically significant in each month of exposure in comparison to the control group (P=0.001) (Fig. 1). In the control group, the highest changes of MN were observed in the second month and there was no significant difference between the months of measurement (P>0.05).
| Group | Month 1 | Month 2 | Month 3 | Month 4 | P Value |
|---|---|---|---|---|---|
| 1 | 11±0.57 | 15.3±3.05 | 21.6±1.15 | 21.6±1.15 | 0.001 |
| 2 | 8.6±0.57 | 11.6±0.57 | 13.6±1.5 | 15.6±3.2 | 0.001 |
| 3 | 7.3±0.57 | 8.6±0.57 | 11.3±0.57 | 14±1 | 0.001 |
| 4 | 7.6±0.57 | 11±1 | 12.3±0.57 | 13.6±2.5 | 0.001 |
| 5 | 6.3±0.57 | 11±1 | 12.3±1.5 | 13.6±1 | 0.001 |
| Control | 3.3±0.5 | 4.3± 0.8 | 3±1 | 4±1 | -------- |
| Variable | Test | Dust Concentration (mg/m3) | Duration of Exposure (month) |
|---|---|---|---|
| Micronucleus (MN) |
r | 0.679 | 0.459 |
| P-Value | 0.001 | P<0.001 |
The statistical analysis indicated that there is a direct and significant correlation between the concentrations of exposure to silica dust in the exposure groups with average number of formed MN (r=0.679). Table 3 presents the relationship between the formed MN and the concentration and exposure time to silica dust.
4. DISCUSSION
Exposure to silica can occur in occupational and industrial environments due to its abundance in earth crust. Silica is widely used in industries and may cause different damages in human body. Recent studies have shown that when silica dust reaches the original lung tissue, they are phagocytosed by lung macrophages; this activates the production of Reactive Oxygen Species (ROS). ROS can cause damage to cell membranes and genetic structures of various organs of the body, especially the lung [25, 26]. The results of this study indicate that dust concentration and exposure time increase the MN formation. There was a direct and significant correlation between the concentration of silica dust and duration of exposure in the exposure groups with the average number of formed MN (P=0.001). Maximum rate of MN formation was observed in the fourth month and in group one which was exposed to higher concentrations than the rest of the groups; minimum MN formation was observed in group 5 and in the first month, furthermore, rate of MN formation was significantly different in all exposure groups in comparison with control groups. Bonassi et al., conducted a study on the frequency of MN of peripheral blood lymphocytes in order to predict cancer risk in which the frequency of MN of peripheral blood lymphocytes was used as a biomarker of chromosomal aberrations in human populations. Their reported results indicated that the frequency of MN in peripheral blood lymphocytes can be a predictor of cancer risk among healthy people [27]. In a study on workers exposed to crystalline silica dust in Turkey, MN test was performed in peripheral blood lymphocytes and in nasal epithelial cells as a target organ in the respiratory tract. The results indicated that the frequency of MN in lymphocytes was two times more, and in nasal cells three times more than people with exposure background to dust [11].
Based on the results, it can be said that the frequency of MN in blood cells is positively associated with exposure time and is an indicator of cumulative genetic damage due to exposure to crystalline silica. In a study in India on the workers of pottery industry who were in chronic exposure to silica, MN frequency and chromosomal aberrations in peripheral blood lymphocytes were two times more than the control group. Furthermore, the rate of chromosomal aberrations in lymphocytes was significantly high. In some workers, respiratory disorders were observed, and risk of increased mutagenic risk in pottery workers exposed to silica dust was reported to be high [28]. In another study in India on construction workers who were in long-term exposure to silica-contained clay dust, a significant increase was observed in MN of blood leucocytes and comet tail length in comparison with the control group. The reported results of this study indicated that long-term occupational exposure to clay during constructions may increase DNA aberrations and hinder their repair [29].
In a study on tunnel construction workers of Italy who were exposed to a combination of chemical material including silica and stone dust, gases, asbestos, and diesel exhaust compared with outdoor areas workers in the same working area. Primary DNA aberration, Sister-Chromatid Exchanges (SCE), and MN of peripheral blood cells were measured. No significant difference was observed in primary DNA aberration and SCE frequency of tunnel workers and control group, while MN had significantly increased in exposure group in comparison with the control group. Since MN in peripheral blood lymphocytes is known as a predictor of cancer risk in the population with healthy people, genotoxic risk may be caused from occupational exposure to different peripheral pollutions during tunnel construction [30].
In some studies, significant correlations between exposure to crystalline silica and risk of death from lung cancer and silicosis have been reported [31-33]. The results of a prospective study by Chen et al., showed that lung cancer due to exposure to silica was the third most common cause of death among miners in China [34]. Finkelstein, in his study, investigated the exposure limit and risk assessment of cancer due to to silica dust, and stated that risk of cancer would be reduced to below 5% if the recommended exposure limit according to NIOSH was a 0.05 milligram per cubic meter [35]. Liu et al., in a cohort study, evaluated the risk of cancer-induced mortality from exposure to silica in 24,000 workers over the past 44 years and reported 542 deaths due to lung cancer [36].
In this study, the mice were exposed to pure silica dust processed in silica mines of Iran and the research was conducted in controlled conditions. Therefore, considering the fact that in this study, the intervention parameters available in industrial environments and labor studies have been eliminated, changes in MN content of blood lymphocytes of mice can be attributed to silica dust.
CONCLUSION
The present study was conducted in controlled laboratory conditions on animals. The results indicated that exposure to silica dust may form significant amounts of MN in blood lymphocytes of male mice BALB/c in comparison with the control group and as exposure to dust concentrations and exposure time increase, variations in MN increase as well. Since MN in peripheral blood lymphocytes is considered as a predictor of cancer risk, and in this study, changes in MN were observed to be caused by exposure to silica dust, further laboratory and environmental studies are required.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by Birjand University of Medical Sciences, Tehran, Iran.
HUMAN AND ANIMAL RIGHTS
The reported experiments are in accordance with the standards of The US Public Health Service's “Policy on Humane Care and Use of Laboratory Animals,” and “Guide for the Care and Use of Laboratory Animals.”
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTERESTS
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


